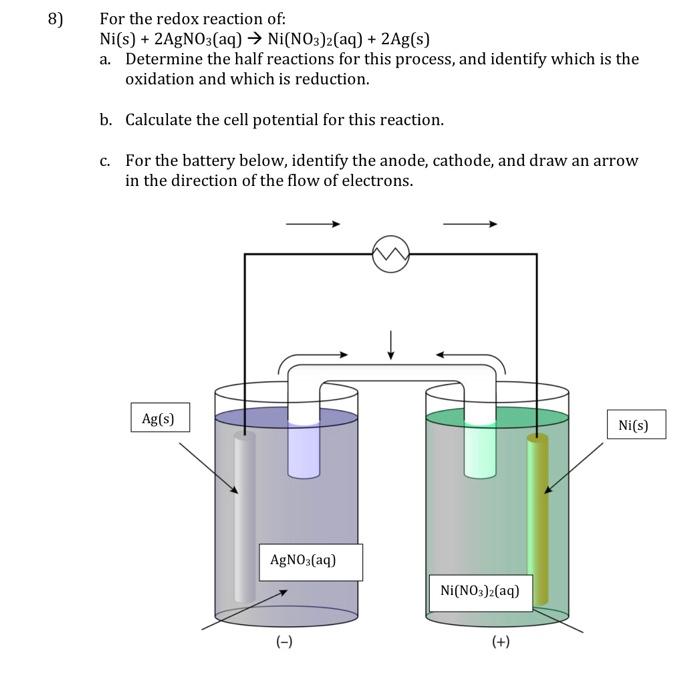
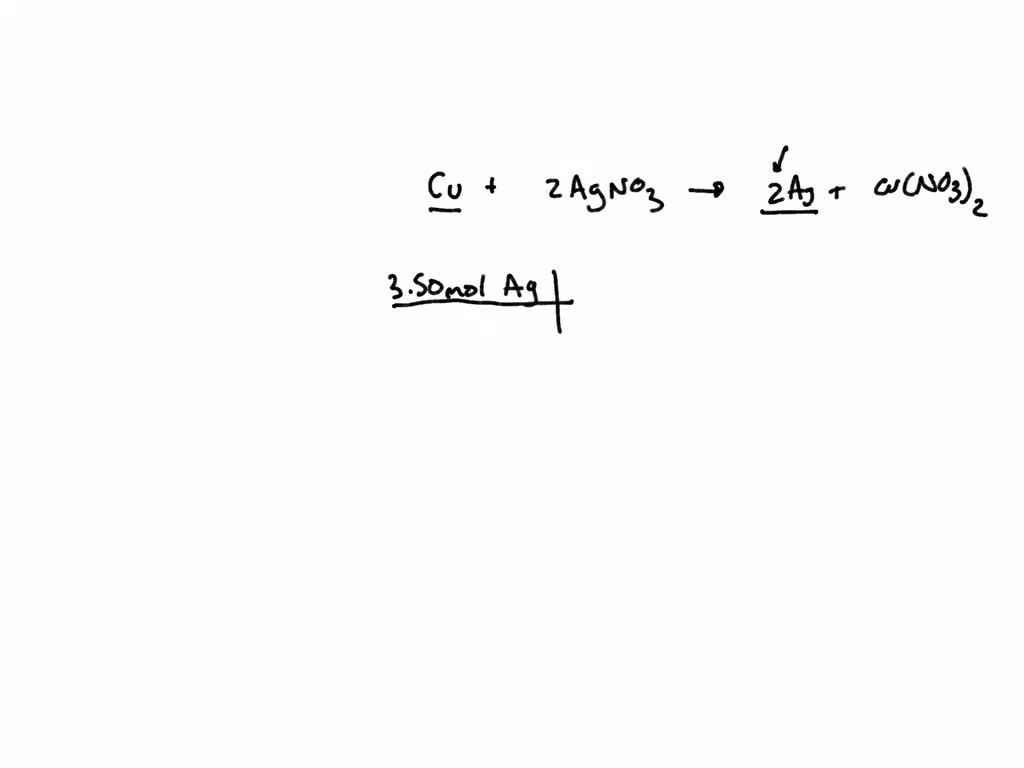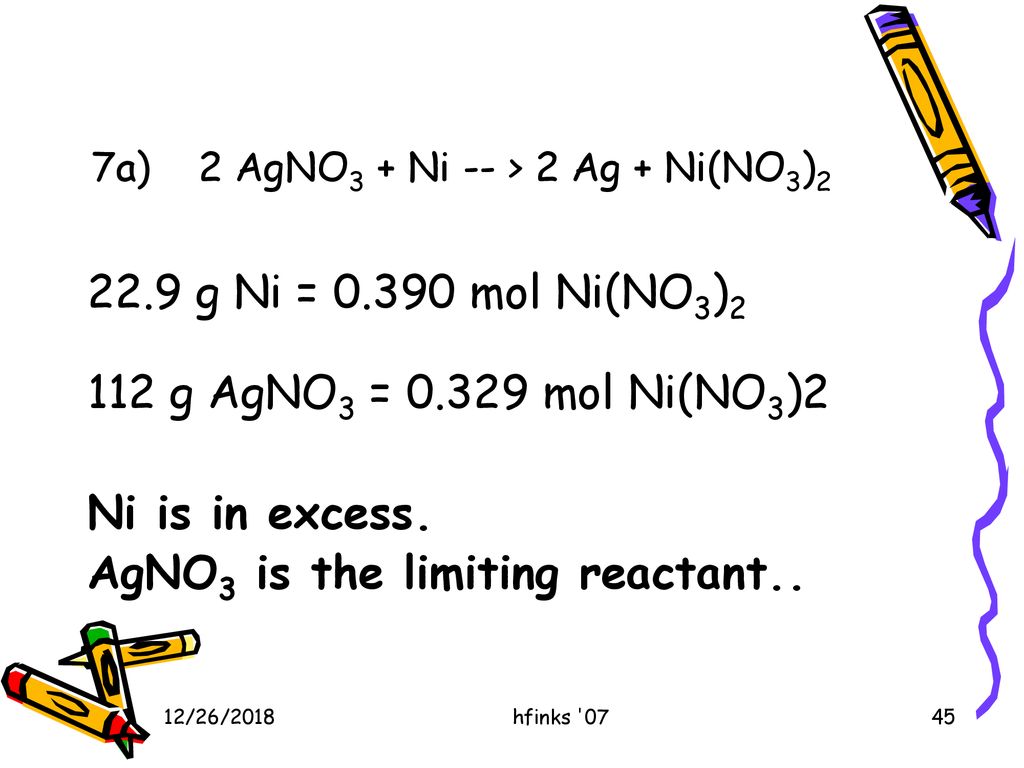Direct Growth of Ag/Ni(OH)2 Composite on Cu Foam by a Modified Galvanic Displacement Reaction Followed by Electrodeposition
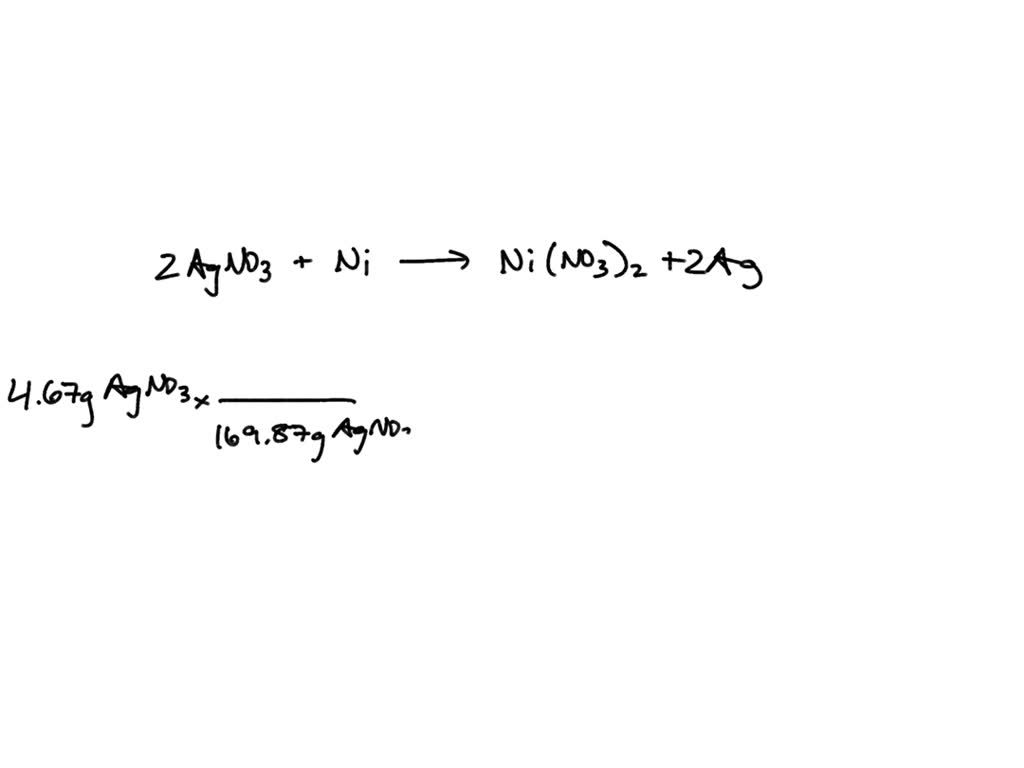
SOLVED: Nickel is a more active metal than silver; therefore, nickel will replace silver ions in a silver nitrate solution to form Nickel (II) nitrate and silver metal. How many grams of

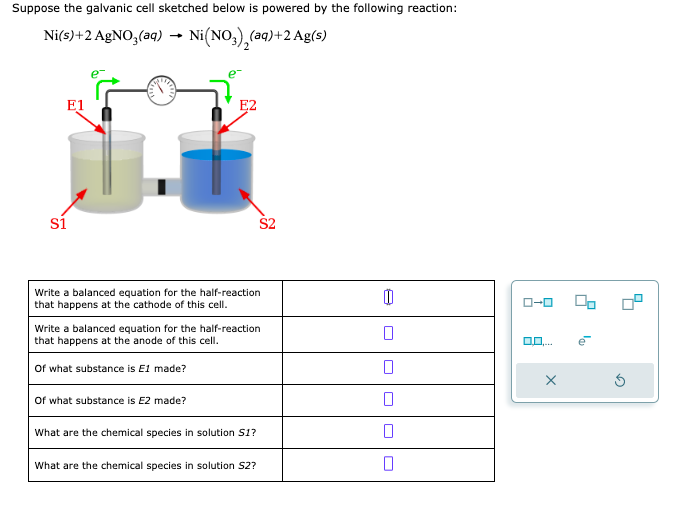
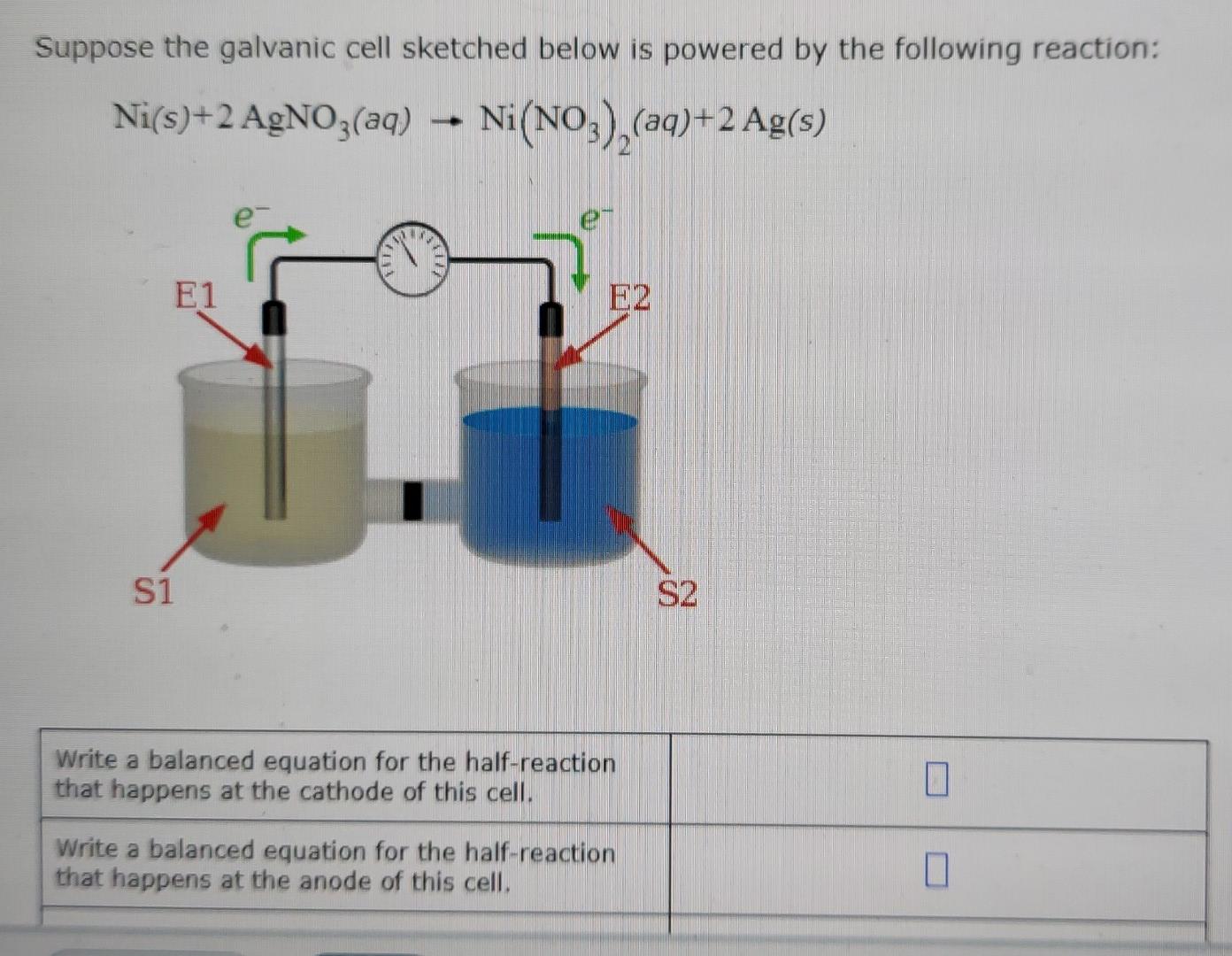
A strip of nickel metal is placed in a 1 molar solution of Ni(NO3)2 and a strip of silver metal - Chemistry - Electrochemistry - 9165083 | Meritnation.com