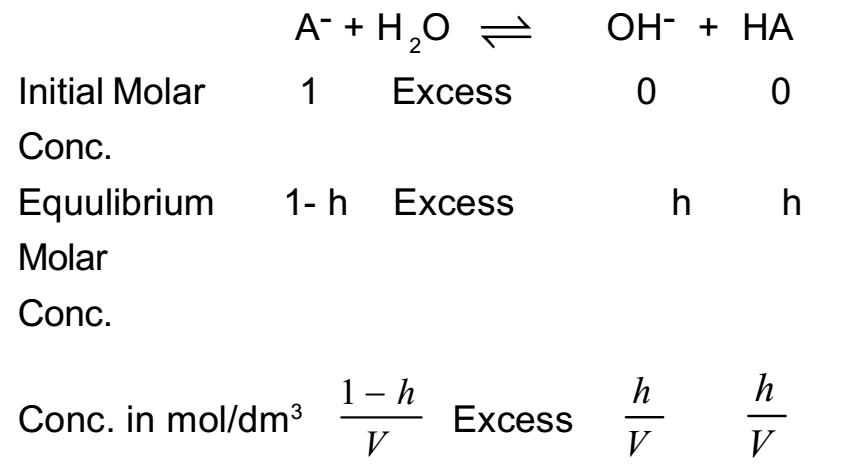One material, multiple functions: graphene/Ni(OH)2 thin films applied in batteries, electrochromism and sensors | Scientific Reports

Metallic Gold-Incorporated Ni(OH)2 for Enhanced Water Oxidation in an Alkaline Medium: A Simple Wet-Chemical Approach | Inorganic Chemistry

Effects of Fe Electrolyte Impurities on Ni(OH)2/NiOOH Structure and Oxygen Evolution Activity | The Journal of Physical Chemistry C

The acid ionziation (hydrolysis) constant of Zn^2 + is 1.0 × 10^-9 (a) Calculate the pH of a 0.001 M solution of ZnCl2 (b) What is the basic dissociation constant of Zn(OH)^+ ?

Metallic Gold-Incorporated Ni(OH)2 for Enhanced Water Oxidation in an Alkaline Medium: A Simple Wet-Chemical Approach | Inorganic Chemistry

Kinetics and mechanism of formation of nickel(II)porphyrin and its interaction with DNA in aqueous medium | SpringerLink

Stability-Enhanced α-Ni(OH)2 Pillared by Metaborate Anions for Pseudocapacitors | ACS Applied Materials & Interfaces

Speciation diagram of Ni 2+ species as a function of solution pH. The... | Download Scientific Diagram

NiSe2/Ni(OH)2 Heterojunction Composite through Epitaxial-like Strategy as High-Rate Battery-Type Electrode Material | SpringerLink

Activity Origins and Design Principles of Nickel-Based Catalysts for Nucleophile Electrooxidation - ScienceDirect

Metallic Gold-Incorporated Ni(OH)2 for Enhanced Water Oxidation in an Alkaline Medium: A Simple Wet-Chemical Approach | Inorganic Chemistry

Nanoflower Ni(OH) 2 grown in situ on Ni foam for high-performance supercapacitor electrode materials - Sustainable Energy & Fuels (RSC Publishing) DOI:10.1039/D1SE01036K

Hydrolysis of Methoxylated Nickel Hydroxide Leading to Single-Layer Ni(OH)2 Nanosheets | Inorganic Chemistry

Nickel hydroxides and related materials: a review of their structures, synthesis and properties | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences