Radical Mechanism of IrIII/NiII-Metallaphotoredox-Catalyzed C(sp3)–H Functionalization Triggered by Proton-Coupled Electron Transfer: Theoretical Insight | CCS Chem

Reaction of Nitrogen‐Radicals with Organometallics Under Ni‐Catalysis: N‐Arylations and Amino‐Functionalization Cascades - Angelini - 2019 - Angewandte Chemie International Edition - Wiley Online Library

Photoredox Ni-Catalyzed Selective Coupling of Organic Halides and Oxalates to Esters via Alkoxycarbonyl Radical Intermediates | CCS Chemistry
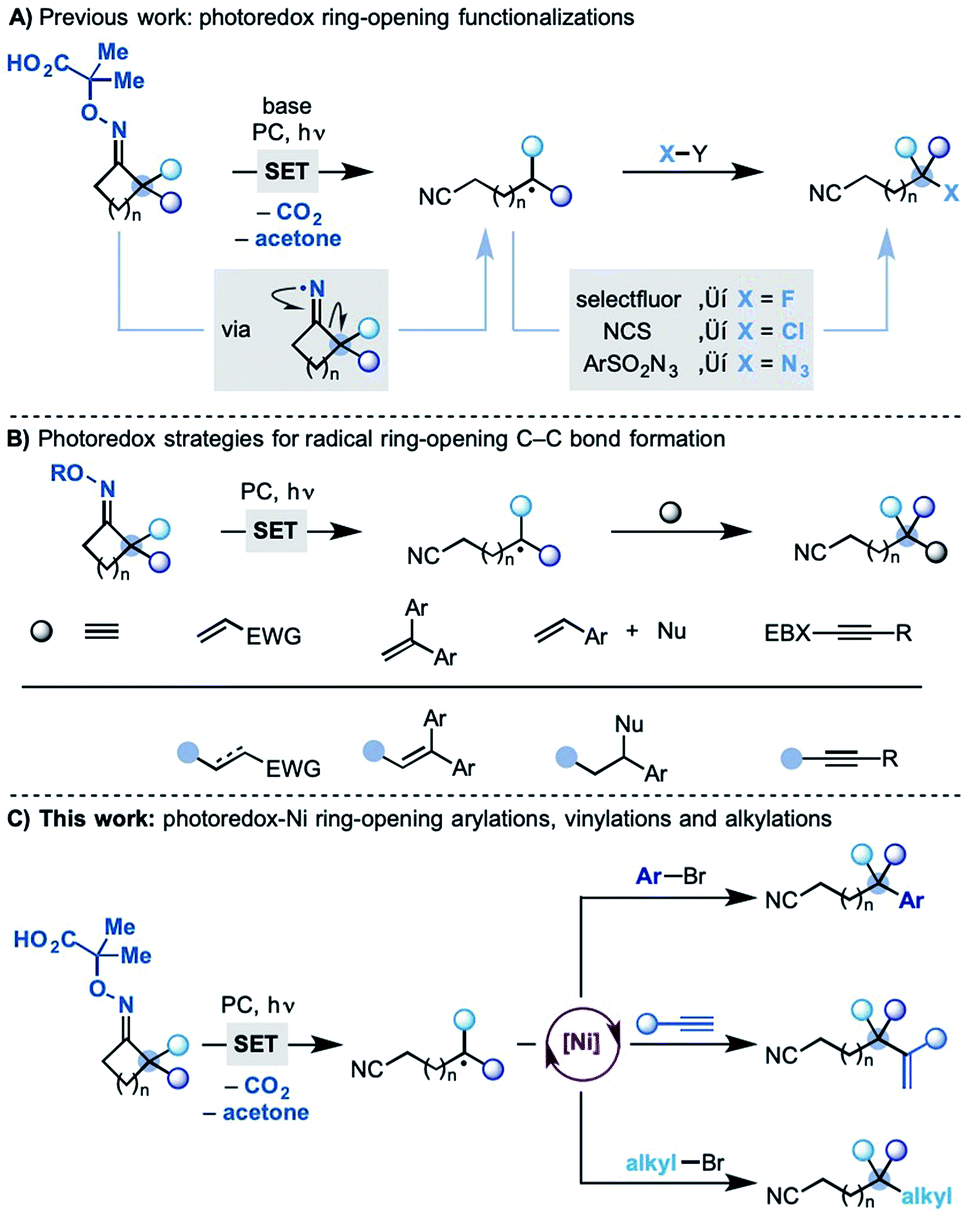
A dual photoredox-nickel strategy for remote functionalization via iminyl radicals: radical ring-opening-arylation, -vinylation and -alkylation cascad ... - Chemical Science (RSC Publishing) DOI:10.1039/C9SC02616A

Ni-catalyzed, enantioselective three-component radical relayed reductive coupling of alkynes: Synthesis of axially chiral styrenes - ScienceDirect

Monovalent Nickel-Mediated Radical Formation: A Concerted Halogen-Atom Dissociation Pathway Determined by Electroanalytical Studies | Journal of the American Chemical Society

Theoretical Study of Ni-Catalyzed C–N Radical–Radical Cross-Coupling | The Journal of Organic Chemistry

Cascade Radical 1,2-N-Shift/C-H Bond Coupling, Halogenation and Selenation Enabled by Cooperative Ni/Diboron Catalysis | Organic Chemistry | ChemRxiv | Cambridge Open Engage

C(sp3)–H methylation enabled by peroxide photosensitization and Ni-mediated radical coupling | Science

Silyl Radical Activation of Alkyl Halides in Metallaphotoredox Catalysis: A Unique Pathway for Cross-Electrophile Coupling | Journal of the American Chemical Society

Theoretical Study of Ni-Catalyzed C–N Radical–Radical Cross-Coupling | The Journal of Organic Chemistry

Photoredox Ni-Catalyzed Selective Coupling of Organic Halides and Oxalates to Esters via Alkoxycarbonyl Radical Intermediates | CCS Chemistry

Free radical addition reactions of benzene with hydrogen methylbenzene chlorination of side-chain with chlorine substitution in methyl group mechanism advanced A level organic chemistry revision notes doc brown

Reaction of Nitrogen‐Radicals with Organometallics Under Ni‐Catalysis: N‐Arylations and Amino‐Functionalization Cascades - Angelini - 2019 - Angewandte Chemie International Edition - Wiley Online Library
![PDF) Hydroxyl radical reactions with the nickel(II) macrocyclic complexes Ni/sup II/Cr, Ni/sup II/(CR-2H), and Ni/sup II/(CR + 4H). Generation of ligand radicals and nickel(III) species. [Gamma radiation] | Larry Patterson - Academia.edu PDF) Hydroxyl radical reactions with the nickel(II) macrocyclic complexes Ni/sup II/Cr, Ni/sup II/(CR-2H), and Ni/sup II/(CR + 4H). Generation of ligand radicals and nickel(III) species. [Gamma radiation] | Larry Patterson - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/53008901/mini_magick20190122-28564-1ja1wi4.png?1548163491)
PDF) Hydroxyl radical reactions with the nickel(II) macrocyclic complexes Ni/sup II/Cr, Ni/sup II/(CR-2H), and Ni/sup II/(CR + 4H). Generation of ligand radicals and nickel(III) species. [Gamma radiation] | Larry Patterson - Academia.edu

IJMS | Free Full-Text | Investigating the Mechanism of Ni-Catalyzed Coupling of Photoredox-Generated Alkyl Radicals and Aryl Bromides: A Computational Study

Intermolecular selective carboacylation of alkenes via nickel-catalyzed reductive radical relay | Nature Communications
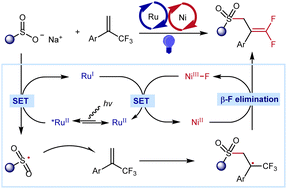
Photo/Ni dual-catalyzed radical defluorinative sulfonylation to synthesize gem-difluoro allylsulfones - Chemical Communications (RSC Publishing)
![IWO NI - Radicals for Jesus [@radical7_4] ft. Samsong [@Samsongfans] - GospelNaija! - Nigerian Gospel Music Promotion and Christian News IWO NI - Radicals for Jesus [@radical7_4] ft. Samsong [@Samsongfans] - GospelNaija! - Nigerian Gospel Music Promotion and Christian News](https://gospelnaija.com/media/com_hwdmediashare/files/1c/ce/ab/6365fd35fcd1f99a0659dd4e892dec24.jpg)
IWO NI - Radicals for Jesus [@radical7_4] ft. Samsong [@Samsongfans] - GospelNaija! - Nigerian Gospel Music Promotion and Christian News

Mechanism of Ni-Catalyzed Oxidations of Unactivated C(sp3)–H Bonds | Journal of the American Chemical Society

The effects of elevated Cd and Ni soil contents on radical scavenger... | Download Scientific Diagram