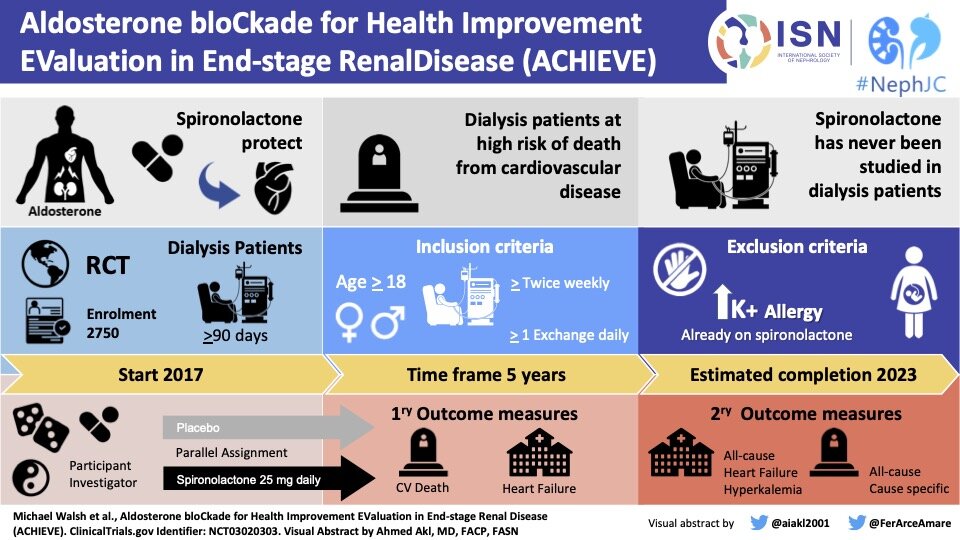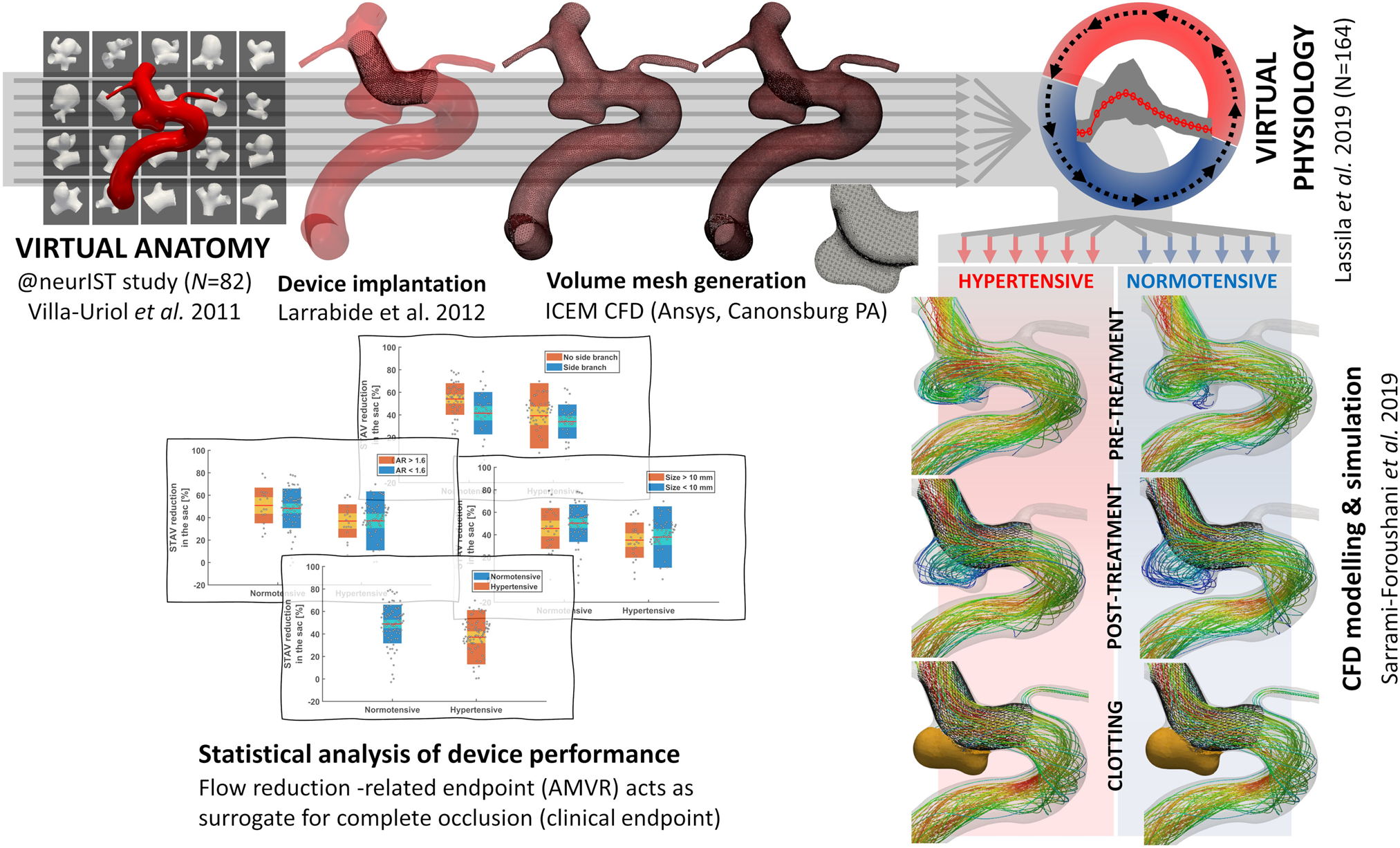
In-silico trial of intracranial flow diverters replicates and expands insights from conventional clinical trials | Nature Communications

Process/Flow or Approaches for determination of Phase 2 trial design... | Download Scientific Diagram
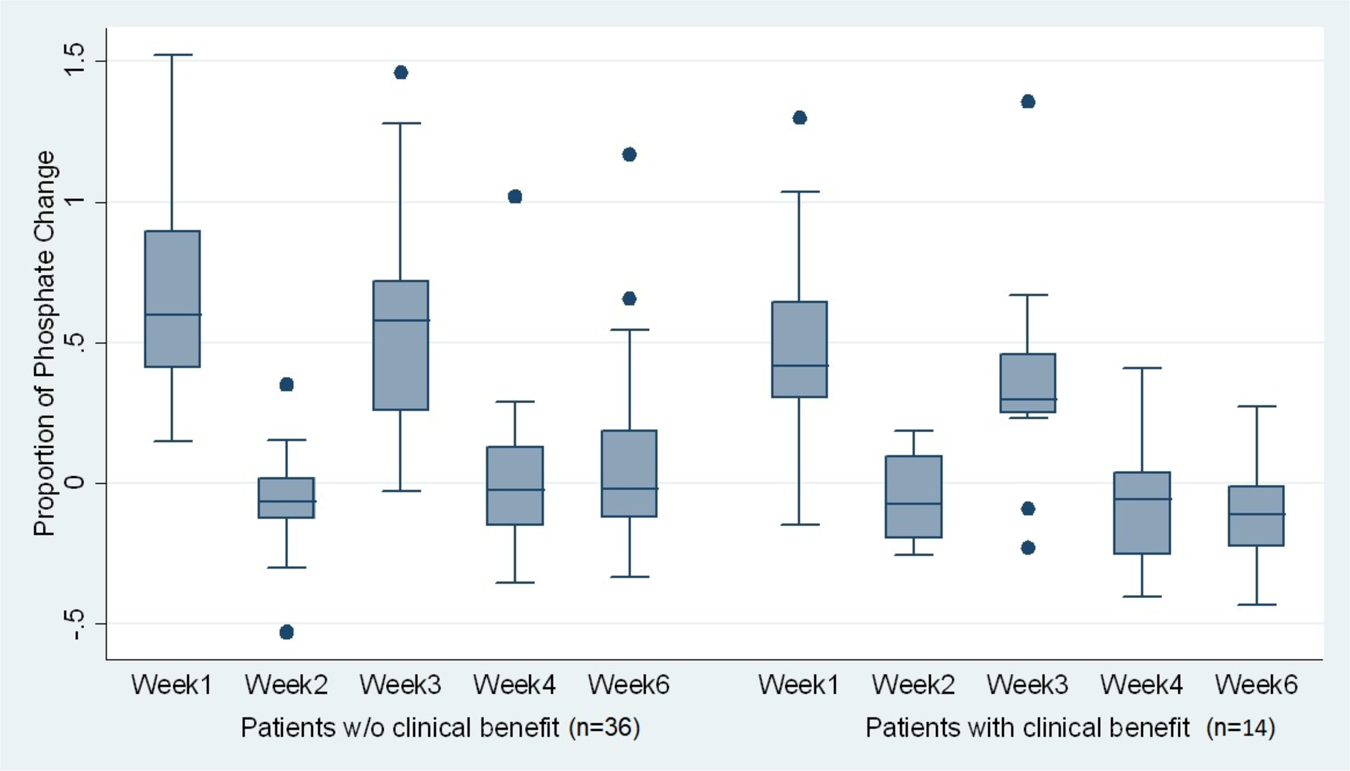
Results of the phase IIa RADICAL trial of the FGFR inhibitor AZD4547 in endocrine resistant breast cancer | Nature Communications

NIA-Funded Active Alzheimer's and Related Dementias Clinical Trials and Studies | National Institute on Aging
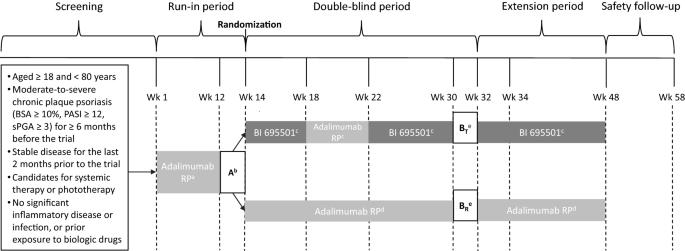
Switching Between Adalimumab Reference Product and BI 695501 in Patients with Chronic Plaque Psoriasis (VOLTAIRE-X): A Randomized Controlled Trial | SpringerLink

study design. Notes: Throughout the run-in phase, all patients received... | Download Scientific Diagram